5 Comments
At the start of the year, we briefly touched on our efforts to address research integrity violations in our 2022 Year In Review. Today we are sharing some more information on the overall trends in research integrity allegations associated with the NIH grants process. I want to note that while we are sharing these aggregate data, NIH does not discuss grants compliance reviews on specific funded awards, recipient institutions, or supported investigators, and whether such reviews occurred or are underway.
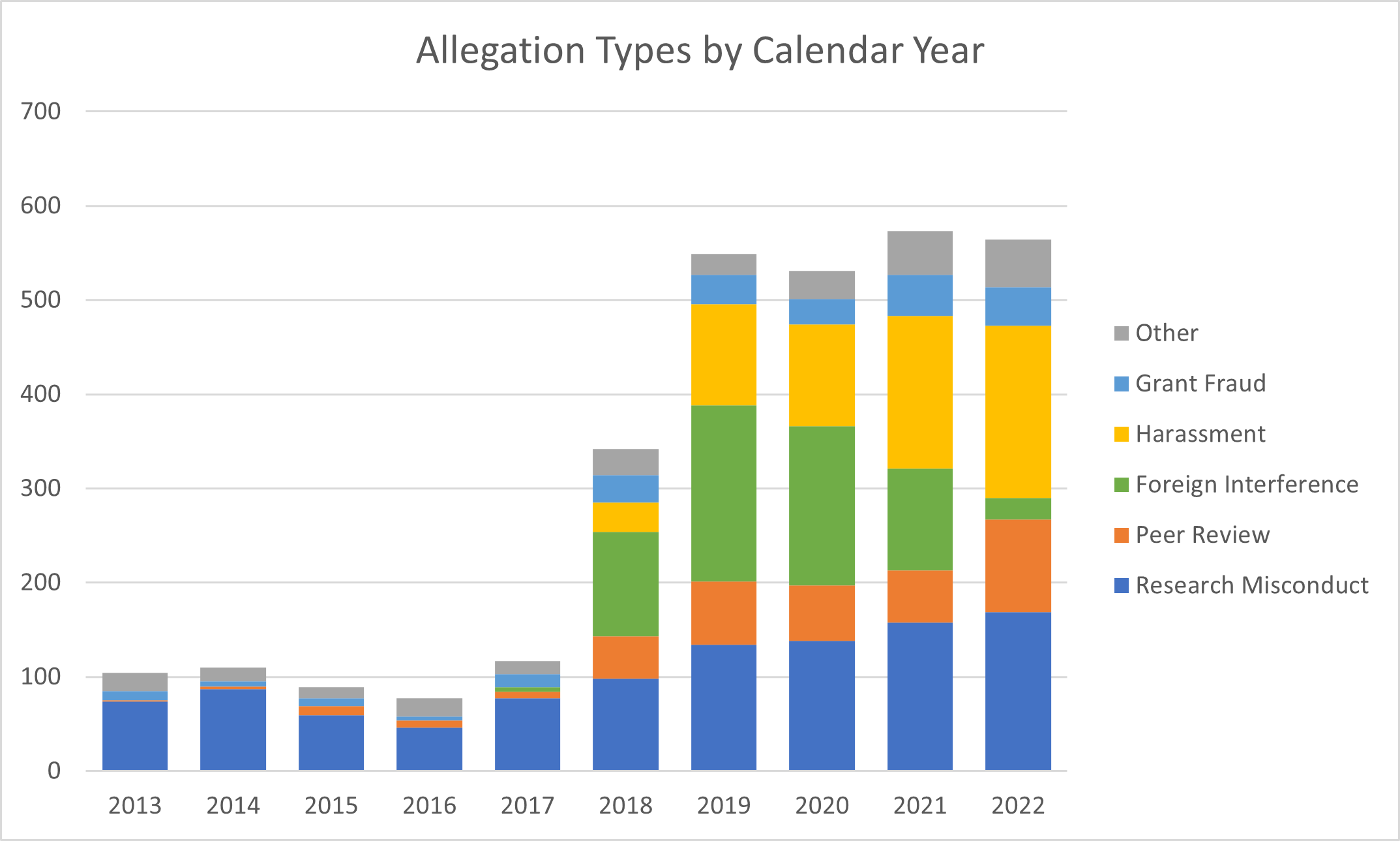
Table 1 shows the total number of new research integrity allegation types that NIH received between calendar years (CYs) 2013 and 2022. These include allegations related to traditional research misconduct and professional misconduct, such as peer review, foreign interference, harassment, grant fraud, and other types. We generally handled an average of 100 violations each year up to around 2017. Over the last five years, however, the numbers rose precipitously.
Table 1: Total Research Integrity Allegation Types: CYs 2013 to 2022
| Calendar Year | Allegations* |
| 2013 | 104 |
| 2014 | 110 |
| 2015 | 89 |
| 2016 | 77 |
| 2017 | 117 |
| 2018 | 342 |
| 2019 | 549 |
| 2020 | 531 |
| 2021 | 573 |
| 2022 | 564 |
* Represents the number of allegation types received each calendar year (not the number of allegations). For instance, an allegation that involves two different types (such as research misconduct and harassment) is counted twice.
Our integrity portfolio broadened greatly around 2018 as professional misconduct became a major focus along with traditional scientific misconduct. Importantly, we also made concerted efforts with the research community over recent years to identify and address integrity issues.
Table 2 breaks down the data presented above by the type of research integrity allegation. Here are the highlights:
- Allegations of research misconduct (fabrication, falsification, and plagiarism) were generally less than a hundred per year, but the numbers started rising in 2019. This increase may be due to our reminder in late 2018 that recipients notify NIH when they find, learn of, or suspect research misconduct that impacts or may impact an NIH-supported project. Check out our webpage on how NIH handles research misconduct allegations for more details.
- Alleged violations of peer review rules increased from fewer than ten in 2013 when we first started tracking them, to nearly a hundred in 2022. This increase is also commensurate with many reminders to the research community about rules and violations, such as sharing and disclosing of confidential peer review materials with unauthorized individuals. Read more about these types of violations on our peer review case studies page.
- Foreign interference allegations associated with the NIH grants process were tracked starting around 2017. The number of these types of allegations peaked around 2019 but have substantially decreased over the past few years (likely related to our outreach and increased awareness in the community of reporting requirements). Data on outcomes of foreign interference allegations are updated semi-annually on our site.
- Allegations and notifications related to harassment (including sexual harassment), discrimination, and hostile work environments have increased substantially since we started tracking these in 2018. This rise in numbers is likely due in part to the heightened awareness and attention about harassment in the scientific workforce, together with our outreach efforts and strengthened recipient notification requirements. See our site for more data on harassment allegations and outcomes.
- The number of grant fraud allegations have increased proportionally with the overall increase in allegation numbers of the years. The NIH Grants Policy Statement shares examples of grant fraud including embezzlement, misuse of property, theft of funds, or false statements. Of note, we refer the grant fraud allegations we receive to the NIH Office of Management Assessment, who is charged with receiving and handling these types of allegations.
- The final category, referred to as “Other”, includes other types of research integrity concerns, such as misrepresentations in grant documents (e.g., false credentials in a biosketch) and intellectual property or patent disputes.
Table 2: Research Integrity Allegations by Type: CYs 2013 to 2022
| Calendar Year | Research Misconduct | Peer Review | Foreign Interference | Harassment | Grant Fraud | Other |
| 2013 | 74 | 1 | 0 | 0 | 10 | 19 |
| 2014 | 87 | 3 | 0 | 0 | 5 | 15 |
| 2015 | 59 | 10 | 0 | 0 | 8 | 12 |
| 2016 | 46 | 8 | 0 | 0 | 4 | 19 |
| 2017 | 77 | 7 | 5 | 0 | 14 | 14 |
| 2018 | 98 | 45 | 111 | 31 | 29 | 28 |
| 2019 | 134 | 67 | 187 | 108 | 31 | 22 |
| 2020 | 138 | 59 | 169 | 108 | 27 | 30 |
| 2021 | 158 | 55 | 108 | 162 | 44 | 46 |
| 2022 | 169 | 98 | 23 | 183 | 41 | 50 |
Figure 1 below is a visual representation of the same CY 2013 to 2022 data as presented in Table 2.
In April 1999 the National Science and Technology Council issued a report on “Renewing the Government-University Partnership.” That report stated as a Guiding Principle “The ethical obligations entailed in accepting public funds and in the conduct of research are of the highest order and recipients must consider the use of these funds as a trust. Great care must be taken to ‘do no harm’ and to act with integrity. The credibility of the entire enterprise relies on the integrity of each of its participants.” NIH strives to exemplify and promote the highest level of integrity, accountability, and stewardship. We appreciate the research community’s continued support and engagement.
If you have any concerns or come across misconduct, we strongly encourage you to report it. Appropriate contacts are available on our Report a Concern page (please also see this NIH Extramural Nexus article).



Thank you for sharing this information. Curious as to whether the allegation numbers represent total of allegations rather than number of cases. E.g. one case may be about 1 or more allegations.
Thank you for reaching out. Please refer to the footnote at the end of Table 1, which notes that every count of an allegation “Represents the number of allegation types received each calendar year (not the number of allegations). For instance, an allegation that involves two different types (such as research misconduct and harassment) is counted twice.”
What is the process once an allegation is made?
Thank you,
This is good information for the university Research Integrity Committee to discuss at the next meeting.
Any Changes in NIH viewpoints or policy changes is important to know.
What is the NIH doing to rectify the careers of those harmed by alleging wrongdoing?