We are pleased to announce that for the first time, data are now available on the age of participants in NIH-supported clinical research. The newly available information on age adds to already reported data on participant sex or gender, race, and ethnicity.
The NIH Inclusion Across the Lifespan (IAL) policy, implemented in response to the 21st Century Cures Act, requires individuals of all ages (including children and older adults) be included in our supported clinical research absent compelling scientific or ethical reasons. Recipients whose projects fall under the policy have been submitting anonymized individual-level data on the sex or gender, race, ethnicity, and age of their participants when they enroll as part of their progress reports (see these posts here for more).
The Research Inclusion Statistics Report now has a table with fiscal year (FY) 2021 data on participant age at enrollment, broken down by NIH Research, Condition, and Disease Classification (RCDC) category. The data are limited to FY 2021 because the first set of funded projects that fell under the IAL policy were submitted on or after January 25, 2019. This snapshot of data, however, provides us a glimpse into the distribution of participant ages across our supported clinical research activities, and serve as a baseline for future analyses (as noted on page 28 of the current NIH-wide Strategic Plan).
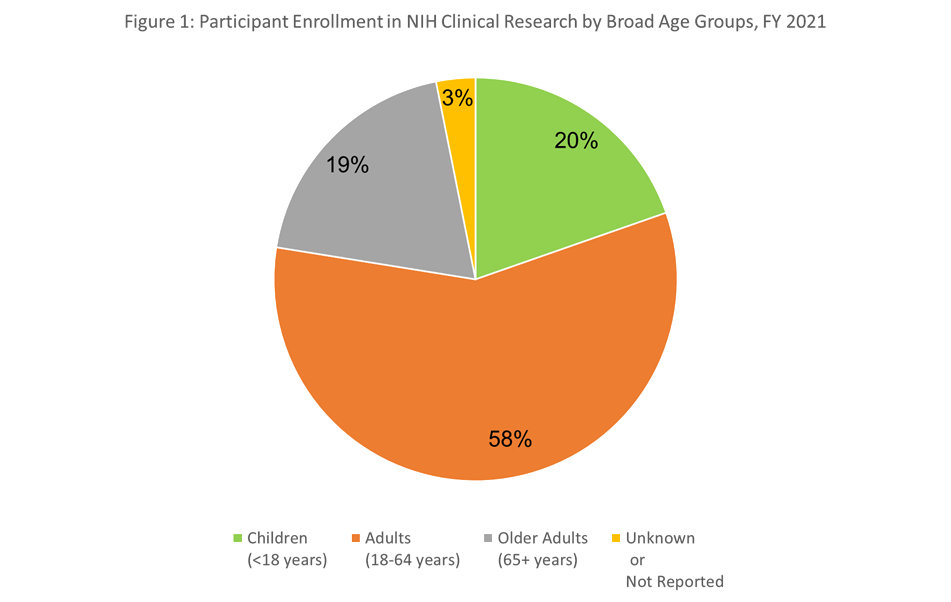
Let’s take a look at the general distribution of participant age across FY2021 NIH supported clinical research that reported age at enrollment. Figure 1 shows the breakdown in broad age categories. Children under 18 years represented 20%, adults between 18 and 65 represented 58%, and adults older than 65 represented 19% of clinical research participants. The age at enrollment was not known or reported for 3% of the participants.
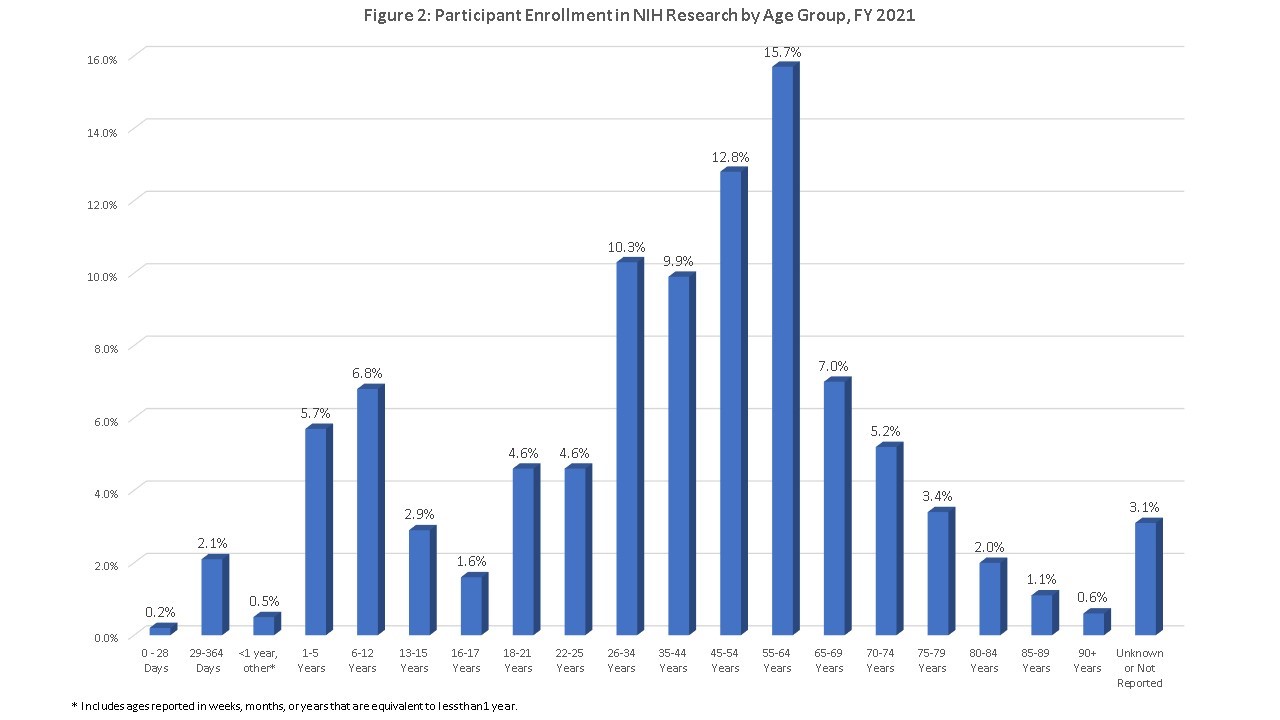
Figure 2 breaks up these broader groups into twenty separate, smaller age groups. Stakeholders at the first IAL meeting in 2017 (see the summary report here) and many comments received in response to a Request for Information (see page 52 for the comments) expressed the importance of these smaller groupings to get a better sense of the distribution of participants at critical points across the lifespan. Of note:
- 42 percent of enrolled children were less than 6 years old, while over a third were aged 6-12 years.
- 23 percent of children were adolescents 13-17 years. We have heard about challenges enrolling adolescents during the IAL2 meeting in 2020 and other publications (see here, here, and here) and will keep continued focus on this age group.
- Among older adults, most participants (63 percent) were less than 75 years of age.
In addition to the new age data being reported by RCDC category, aggregate data on sex or gender, race, and ethnicity of participants are also available in the recently released FY 2019-2020 Biennial Report of the Advisory Committee on Research on Women’s Health. The report highlights NIH’s efforts to monitor adherence to the NIH policy on the inclusion of women and racial and ethnic minorities as subjects in clinical research.
We hope these age-related data, and the updates to them going forward, will be helpful to get a better understanding of who is participating in NIH-supported clinical research.



0 Comments